How to Use Test Cups for Accurate Drug Screening
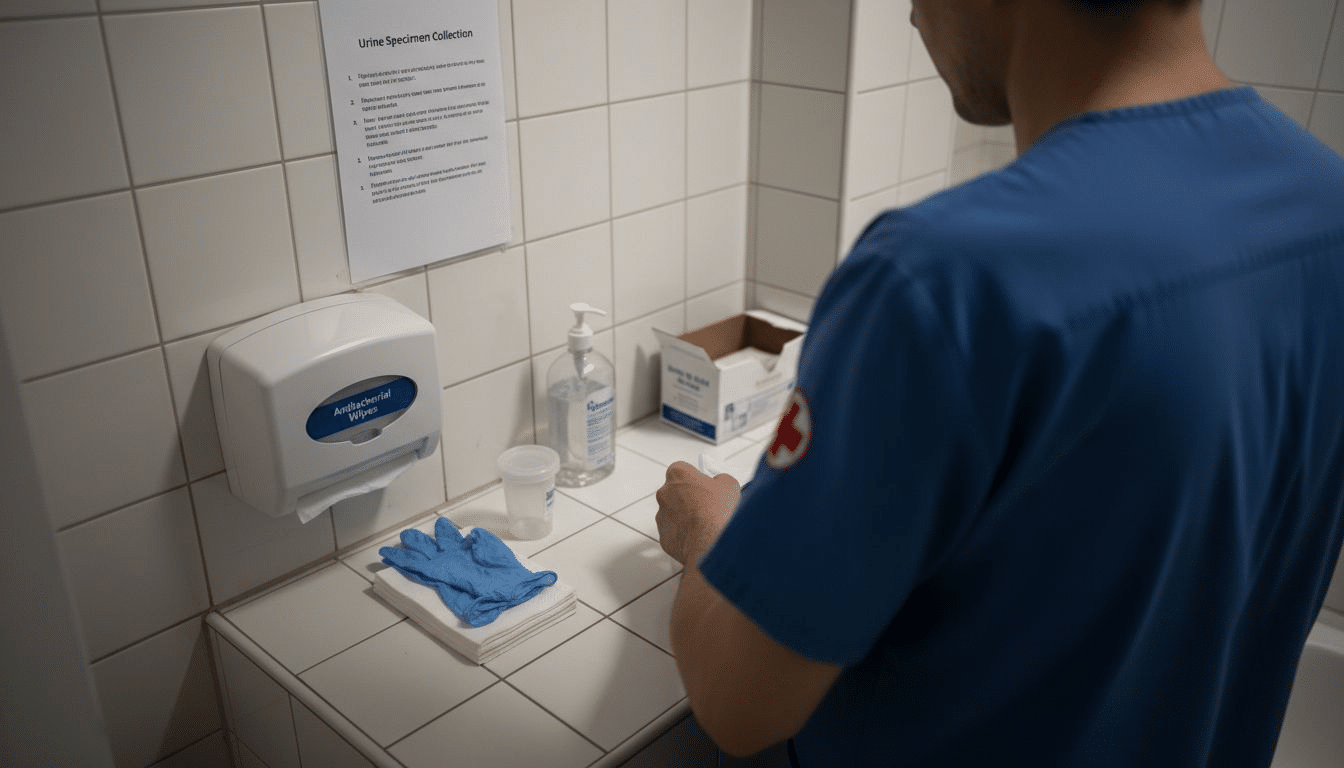
When accuracy in drug screening is non-negotiable, every step in the process matters. Clinical teams across the United States recognize that even minor lapses in preparation can threaten the integrity of compliance monitoring. Organizing your testing materials on a clean surface and using a sterile collection kit forms the backbone of reliable screening, protecting both your patients and your facility’s reputation. This guide details simple, practical steps to help your staff deliver consistent, trustworthy results every time.
Table of Contents
- Step 1: Prepare All Testing Materials
- Step 2: Collect The Urine Sample Correctly
- Step 3: Secure And Label The Test Cup
- Step 4: Perform The Drug Test Procedure
- Step 5: Interpret And Document The Test Results
Quick Summary
| Key Takeaway | Explanation |
|---|---|
| 1. Gather All Testing Materials | Organize and prepare urine collection kits to ensure cleanliness and prevent contamination before the test. |
| 2. Use Proper Collection Techniques | Instruct the individual to use clean-catch midstream collection to minimize the risk of sample contamination. |
| 3. Secure and Label Sample Correctly | Seal and accurately label the sample cup to create a clear chain of custody and maintain test integrity. |
| 4. Follow Testing Protocols Precisely | Adhere to the specific testing procedures to ensure reliable results and document them accurately after testing. |
| 5. Confirm Positive Results with Laboratory Tests | Always send any positive screenings to a certified lab for confirmation to ensure test accuracy and validity. |
Step 1: Prepare All Testing Materials
You’re about to begin a drug screening process, and getting your materials organized is the foundation for accurate results. Before you collect any samples, you need to gather and arrange everything on a clean surface where you’ll conduct the test. This prep work takes just a few minutes but makes a significant difference in preventing contamination and ensuring the integrity of your screening.
Start by unpacking your urine collection kit, which typically includes a sterile urine collection cup with a clearly marked fill line, disposable gloves, and a plastic bag for the sealed specimen. Check that the cup is empty and genuinely sterile before you begin. Lay out all your materials on a clean, flat surface away from any potential sources of contamination. Before touching anything related to the sample collection, put on your disposable gloves. This simple step protects both you and the integrity of the test. According to CLSI guidelines for collection materials, having appropriate labeling tools and clear instructions within reach ensures you can maintain consistency throughout the process. Organize your materials in the order you’ll use them, and keep them within arm’s reach so you’re not fumbling around or leaving your station mid-process. If you’re running multiple screenings, prepare separate kits for each individual rather than reusing materials.
Once everything is arranged and you’re gloved up, you’re ready to move forward with the actual collection. The next step involves communicating clear instructions to the person being tested and ensuring they understand the process before beginning.
Here’s a summary of key materials and their roles during urine drug screening preparation:
| Material | Purpose | Prevents |
|---|---|---|
| Sterile collection cup | Holds urine sample | Sample contamination |
| Disposable gloves | Protects staff and sample | Skin contact, infection |
| Plastic specimen bag | Secures sealed sample | Tampering or spills |
| Labeling tools | Identifies specimen | Sample mix-ups |
| Instructions | Guides procedure | Human error |
Pro tip: Set up your testing station in a private bathroom or designated area away from distractions, and verify that your collection cup hasn’t been opened or tampered with before you begin, as even a small breach can compromise test accuracy.
Step 2: Collect the Urine Sample Correctly
Now that your materials are organized and you’re ready to proceed, the actual collection process requires precision and attention to detail. The way the sample is collected directly impacts test accuracy, so understanding the proper technique matters more than you might think. This step involves guiding the person being tested through a clean-catch midstream collection, which prevents contamination and ensures reliable results.
Start by instructing the individual to wash their hands thoroughly and clean their genital area with soap and water. They should then begin urinating into the toilet for the first few seconds to flush out any bacteria or debris from the urethra. Once the stream has started, they’ll catch the remaining urine midstream into the sterile cup until it reaches the fill line marked on the container. This midstream collection technique is the gold standard for drug screening because it minimizes contamination from skin cells and external sources. The person being tested should avoid touching the inside of the cup or allowing any external substances to make contact with the specimen. Once the cup reaches the fill line, they should finish urinating into the toilet. Immediately after collection, the cup must be securely capped. If the outside of the cup has become wet or contaminated, wipe it clean with a paper towel while keeping the cap secure. The sample should be submitted immediately for testing to maintain its integrity and accuracy.
After the sample is collected and capped, you’ll move into the documentation and labeling phase. Proper chain of custody procedures at this stage protect the validity of your results and maintain the legal standing of the test.
Pro tip: Ensure the person being tested understands the entire process before they enter the collection area, and remind them not to rush, as slower, more controlled collection reduces the chance of spillage or contamination that could compromise the test.
Step 3: Secure and Label the Test Cup
With the sample successfully collected, your next responsibility is to secure it properly and document all relevant information. This step establishes the chain of custody and protects the integrity of your test results. Proper sealing and labeling are not optional procedures but critical safeguards that ensure your screening remains legally valid and tamper-proof.
Begin by ensuring the cup’s lid is tightly sealed. Screw the cap on firmly until it sits snugly against the container, creating a secure fit that prevents any leakage or accidental opening. If you notice any wetness on the exterior of the cup, use a clean paper towel to dry it completely before proceeding. Once the cup is sealed, you’ll apply the label with the donor’s identifying information. This label must include the individual’s name or identification number, the date of collection, and the time the sample was collected. The accuracy of this documentation cannot be overstated because it directly connects the specimen to the specific person being tested. Both you and the person being tested should sign or initial the label to verify that the correct sample has been properly documented. This signature process is your proof of chain of custody, establishing that no tampering has occurred and creating a documented record that protects everyone involved. According to tamper-evident sealing procedures, the specimen must be placed into a locked container or secure storage area immediately after labeling. Never leave an unsealed or improperly labeled cup unattended, as this creates liability and compromises the validity of your testing program.
Once your cup is sealed, labeled, and stored securely, you’re ready to submit it for analysis or store it according to your facility’s protocols. The documentation you’ve completed becomes the official record of this test.
Pro tip: Use waterproof labels or permanent marker to ensure your labeling information remains legible even if moisture accumulates, and always maintain a duplicate copy of the collection documentation in your facility’s records for accountability purposes.
Step 4: Perform the Drug Test Procedure
You’re now ready to execute the actual testing process. This is where your preparation pays off, and where accuracy depends on following the proper protocol. The drug test using your test cup involves applying the specimen to the cup’s testing strips and waiting for results to develop. Understanding each stage of this process ensures you capture reliable data that your facility can trust and act upon.
Begin by reviewing the specific instructions that came with your test cup, as different manufacturers may have slight variations in their procedure. Most modern test cups use immunochromatography competitive assays applied directly to the collected urine specimen. Remove the sealed cup from storage and inspect it one final time to ensure the label is intact and the cap is secure. Open the cup carefully and apply the urine sample to the designated testing wells or strip areas on the inside of the cup, following the manufacturer’s specific volume requirements. Do not overfill or underfill, as either situation can produce inaccurate results. Once the specimen makes contact with the testing strips, a chemical reaction begins that will reveal the presence or absence of drug metabolites in the urine. Set the cup aside in a clean, flat location and start timing according to the instructions. Most test results become visible within about five minutes, though you should wait the full recommended time before reading the cup for accuracy. During this waiting period, avoid moving or jostling the cup, as physical disturbance can affect the reliability of the test results.
When the appropriate time has elapsed, examine the results according to the cup’s indicator windows. One line indicates a negative result, while two lines indicate a positive screening. Document these results immediately and clearly in your testing records. Any positive screening result requires confirmatory testing at a certified laboratory using advanced methods like gas chromatography or liquid chromatography to confirm the initial finding and ensure absolute accuracy before taking any action based on the results.
Pro tip: Train all staff members to read results within the exact timeframe specified by the manufacturer, as results read too early or too late can be misinterpreted, and always photograph or document borderline results before they fade to create a clear record of your findings.
Step 5: Interpret and Document the Test Results
You’ve completed the test and now you’re looking at the results. This final step requires careful attention because how you interpret and record what you see will guide your next decisions. Documentation creates the official record of your screening and ensures that results are communicated accurately to relevant parties within your rehabilitation facility.
Start by carefully examining the result window of your test cup. A single line appearing in the control region only indicates a negative result, meaning the individual tested negative for the substances screened. Two lines, one in the control region and one in the test region, indicate a positive screening result. However, understand that presumptive positive results require confirmatory testing before you take any definitive action. Initial screening tests can occasionally produce false positives, which is why confirmation through laboratory methods like gas chromatography-mass spectrometry is essential for accuracy. Write down your observations immediately while the cup is in front of you. Record the specific substances tested, the date and time of testing, and the exact results you observed. Include any relevant details such as the condition of the specimen, whether the sample appeared normal or showed signs of adulteration, and any unusual circumstances surrounding the collection. Your documentation should also note whether this is an initial screening or a follow-up test, and it must be signed or initialed by you as the testing administrator. When documenting results, consider factors that might affect interpretation, such as prescription medications the individual may be taking that could produce positive results for certain substances. For rehabilitation staff managing patient compliance, this contextual information matters greatly when discussing results with patients or referring them for confirmatory testing. Create a clear, organized record that another staff member could read and immediately understand what happened during the test and what the findings were.
Once results are documented, any positive screening must be sent to a certified laboratory for confirmatory testing. Negative results can typically be filed in the individual’s records according to your facility’s retention policies. Clear, thorough documentation protects your facility legally and ensures that clinical decisions are based on accurate information.
This table outlines differences between initial urine drug screening and confirmatory laboratory testing:
| Test Type | Purpose | Method | Result Reliability |
|---|---|---|---|
| Screening Test | Preliminary detection | Immunochromatography, test cups | Moderate, rapid |
| Confirmatory Test | Verify positives | Gas or liquid chromatography | High, laboratory-based |
Pro tip: Maintain a separate testing log with all results, and always keep documentation organized chronologically by patient, making it easy to track trends in compliance or identify patterns that might warrant additional interventions or adjustments to your monitoring program.
Ensure Precise Drug Screening with Trusted Test Cups and Supplies
Accurate drug screening depends on proper collection, handling, and testing procedures as outlined in the article “How to Use Test Cups for Accurate Drug Screening.” Key challenges include avoiding sample contamination, securing proper labeling, and interpreting results within strict timeframes. You want reliable tools that simplify these critical steps and protect the integrity of your testing program while reducing human error.
At 12 Panel Now, we understand the importance of sterile, easy-to-use test cups and components designed to support the chain of custody protocols you rely on. Whether you need sterile collection cups, labeling materials, or complete drug test kits, our high-quality supplies empower healthcare professionals, rehabilitation staff, and workplace testers to perform screenings with confidence. Learn more about the products and answers to common questions in our Drug Test category and deepen your knowledge through our Complete Drug Test Knowledge Base.
Don’t leave your drug screening accuracy to chance. Visit 12 Panel Now today to find reliable drug testing supplies tailored to your needs and secure your testing program’s success with tools proven to support accurate, tamper-proof results.
Frequently Asked Questions
How should I prepare my testing materials for drug screening?
To prepare your testing materials, gather all components like the sterile urine collection cup, disposable gloves, and labeling tools on a clean surface. Organize them in the order of use and put on your gloves before handling any materials to prevent contamination.
What is the correct way to collect a urine sample using a test cup?
The correct way to collect a urine sample involves instructing the individual to start urinating into the toilet, then catch the midstream urine in the sterile cup. Make sure to only fill the cup to the marked fill line to ensure accurate results.
How do I secure and label the test cup after collecting the sample?
After collecting the sample, tightly seal the cup’s lid and dry any moisture on the exterior. Then, apply a label with the donor’s identifying information, including their name and the date of collection, to maintain a clear chain of custody.
What steps do I take to perform the drug test using the test cup?
To perform the drug test, review the manufacturer’s instructions, open the sealed cup, and apply the urine sample to the designated testing wells. Wait the recommended time before checking the results, and document them immediately for accurate record-keeping.
How do I interpret and document the results of the drug test?
Interpret the results by checking the indicator windows on the cup; one line means negative, while two lines indicate a positive result. Write down your observations with specifics about the substances tested and sign the documentation to confirm the results and protect the integrity of the test.
Recommended
- What Are Specimen Cups Used For? A Complete Guide – 12PanelNow | 12 Panel Drug Test | Free Shipping
- What Is A Drug Test Cup? – 12PanelNow | 12 Panel Drug Test | Free Shipping
- The Essential Guide to the Role of Drug Test Cups – 12PanelNow | 12 Panel Drug Test | Free Shipping
- 5 Best Drug Test Cups for Workplace Screening – 12 Panel Now
- Hair Analysis Test 2025: Discover Your Health Insights | MyHair

