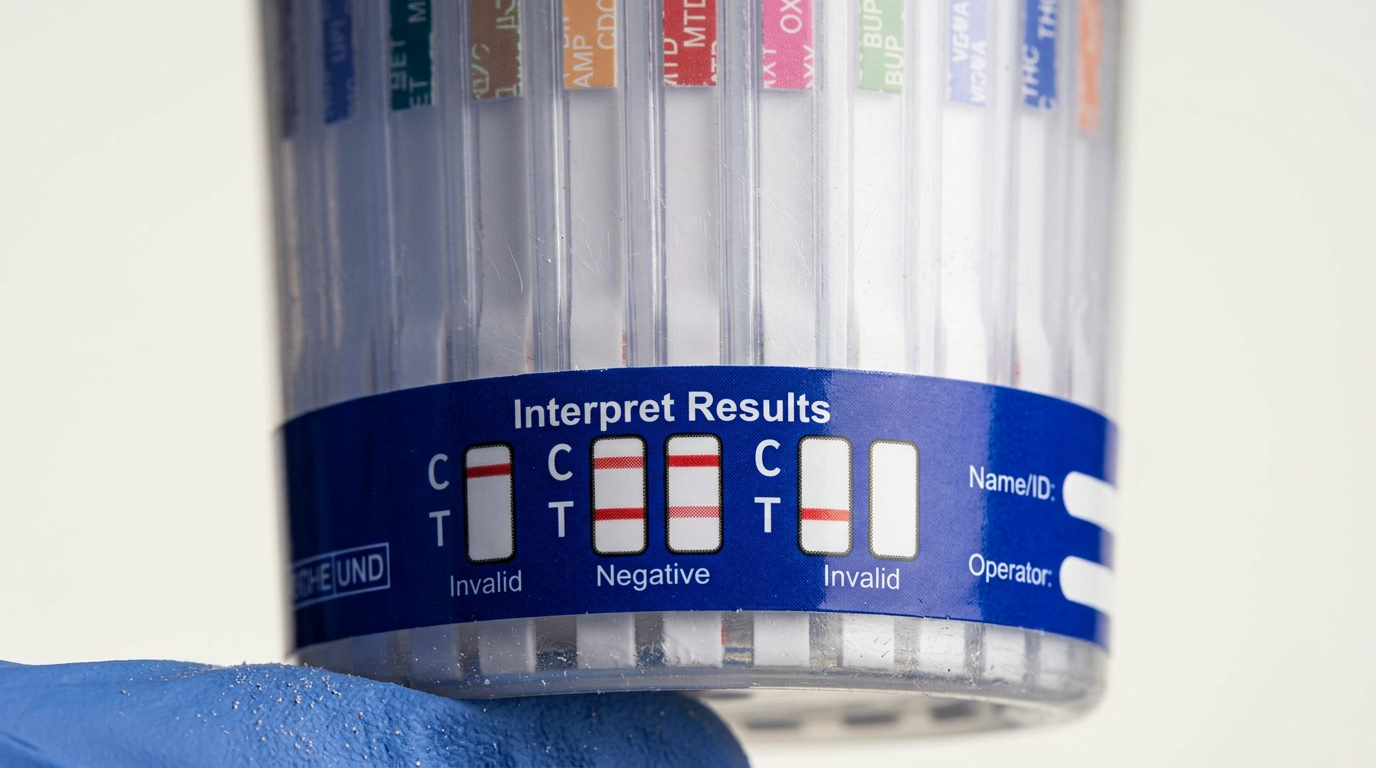
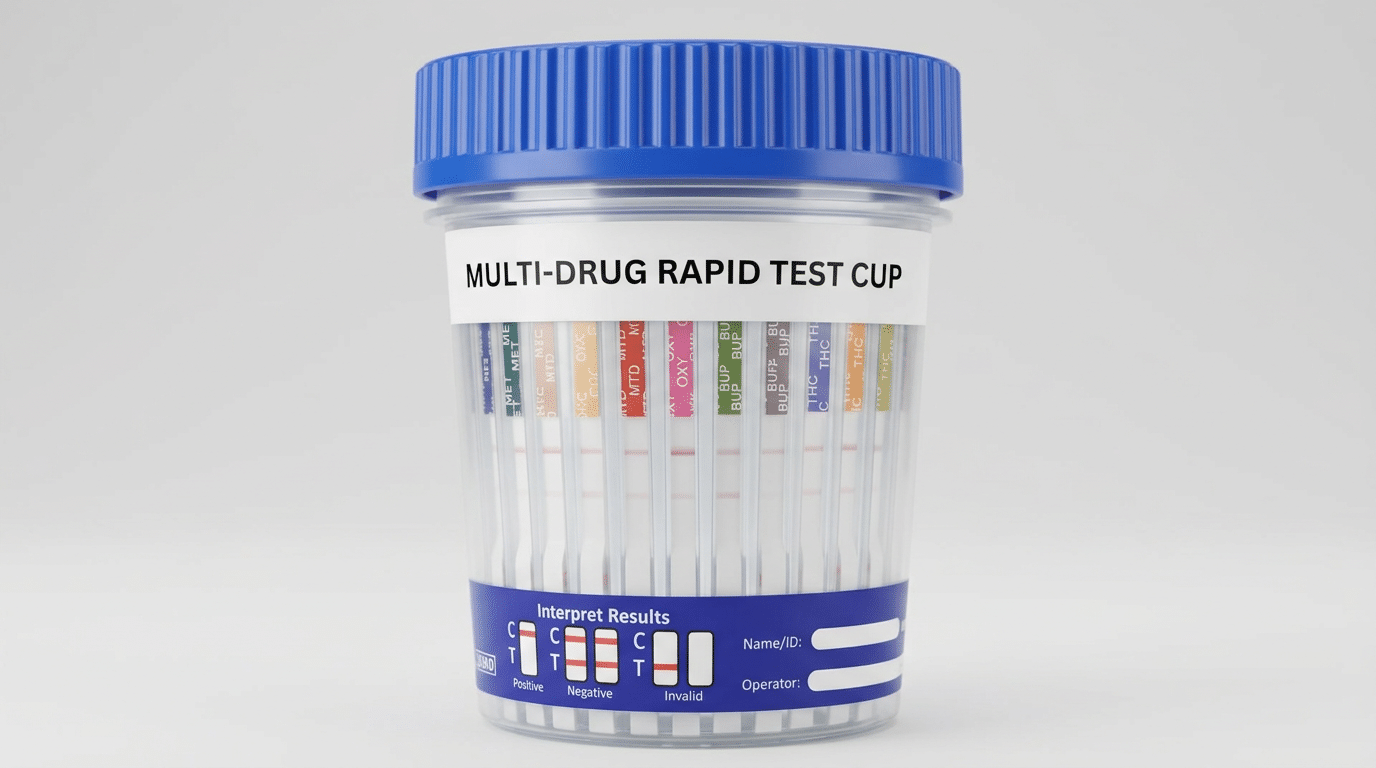
Are Multi-Panel Drug Test Cups FDA-Cleared for ETG/Fentanyl?
When you’re buying multi-panel drug test cups for a clinic, workplace, or monitoring program, the question usually comes down to this: is the exact cup you’re ordering FDA-cleared for professional screening? The answer depends on the specific device and the way it’s labeled. Here’s how FDA clearance applies to rapid urine drug tests, where ETG and fentanyl fit, and what to confirm before you buy.
First: what “FDA-cleared” actually means for drug test cups
“FDA-approved” gets used as a blanket term, but most rapid drug screens don’t follow the same approval pathway as prescription drugs. Many point-of-care tests are reviewed through the FDA’s 510(k) process. When the FDA clears a device through 510(k), it has determined the device is substantially similar to a legally marketed predicate device for the manufacturer’s stated intended use.
That intended use language is the key. FDA clearance is tied to a specific use case, a specific specimen type, and specific analytes. So “FDA-cleared” isn’t a general badge you can apply to any version of a cup—it’s something you verify for the exact configuration you plan to use.
What you should expect from a real FDA clearance claim
If a seller says a multi-panel cup is FDA-cleared, you should be able to confirm it with documentation—not just a product title. A solid claim typically includes a 510(k) number or enough detail to locate the device in FDA records.
- Device identity: a specific brand/model name that matches the documentation.
- Regulatory pathway: FDA 510(k) clearance (not a vague “FDA registered” statement).
- Intended use: screening language that matches how you plan to use it, including specimen type (urine).
- Analyte menu: the exact panels included in the cleared configuration.
Are multi-panel cups with ETG and fentanyl FDA-cleared?
Sometimes. Sometimes not. Multi-panel cups come in countless versions. One configuration may be FDA-cleared for certain drug classes, while another (even if it looks similar) may be sold for forensic, workplace, or at-home use without FDA clearance. You can’t assume clearance just because a seller offers other cleared products or because the cup resembles a cleared device.
ETG and fentanyl also raise the stakes. They’re common in modern monitoring programs, and they’re easy to misunderstand if you rely on the wrong device or the wrong interpretation. Treat any “ETG/Fentanyl FDA-cleared” claim as something you confirm at the product level.
Why ETG and fentanyl raise the “verify it” stakes
ETG screening looks for a metabolite associated with recent alcohol exposure. Programs use it when alcohol abstinence is part of the requirement and when a longer lookback window matters. Fentanyl screening targets a synthetic opioid that’s monitored closely in high-risk clinical and safety-sensitive settings. With both analytes, errors in selection or interpretation can create real consequences.
Rapid cups can still be a practical tool. The point is to start with regulatory status, then make sure you have a clear plan for how you handle non-negative results.
Professional screening: what “professional use” should mean to you
When buyers say “professional screening,” they usually mean two things: the test fits their workflow (workplace, treatment, probation, clinical), and it holds up under audits, policies, and documentation expectations.
Regulatory status matters, but procedures matter just as much. A professional program needs a defined process for collection, read time, documentation, reporting, and confirmatory testing.
Screening vs confirmation: the part buyers get wrong
Most rapid urine cups are screening tests. Screening helps separate likely negatives from results that need follow-up. Confirmatory testing—typically performed in a laboratory—identifies compounds with higher specificity and helps rule out false positives caused by cross-reactivity.
If your program can trigger high-impact outcomes (employment decisions, legal consequences, treatment discharge), treat cup results as preliminary and send non-negative results through your required confirmation process.
How to verify FDA clearance for a multi-panel cup (without guessing)
To determine whether a specific multi-panel cup that includes ETG and fentanyl is FDA-cleared, you need to match the exact product to the exact clearance documentation. Close isn’t good enough here.
Use this quick verification checklist
- Ask for the 510(k) number for the exact device (not “our tests are FDA cleared”).
- Match the device name on the clearance record to the product you’re buying.
- Confirm the analytes listed in the clearance match your panel list, including ETG and fentanyl if required.
- Confirm specimen type is urine and the device type matches (cup vs dip card vs strip).
- Confirm the intended use aligns with your setting and policy requirements.
- File the documentation for audits, inspections, and policy reviews.
Don’t confuse “FDA registered” with “FDA cleared”
Some listings lean on phrases like “FDA registered facility” or “FDA registered.” That typically refers to establishment registration and device listing. It does not mean the FDA reviewed performance the way it does for 510(k) clearance. If your requirement is “FDA-cleared,” you’re looking for 510(k) support.

What about CLIA waiver? Does it matter for cups with ETG/Fentanyl?
CLIA waiver matters when testing is performed in settings regulated under CLIA. A CLIA-waived test is authorized for use in waived environments because it meets criteria for simplicity and low risk of incorrect results when used as directed. CLIA waiver and FDA clearance are related but separate concepts.
If you operate in a physician office, urgent care, or treatment facility, ask whether the exact device is CLIA-waived and follow your site’s CLIA responsibilities. If you’re running workplace testing outside clinical care, CLIA may not apply the same way, but internal policy and state requirements still do.
Common buying questions for ETG/Fentanyl multi-panel cups
“If it’s FDA-cleared, does that mean it’s accurate enough to skip confirmation?”
No. FDA clearance doesn’t eliminate the need for confirmation when policy requires it or when results can lead to serious consequences. Screening tests can produce false positives and false negatives. Confirmation protects both the individual being tested and the integrity of your program.
“Will ETG always mean someone drank alcohol?”
ETG indicates alcohol exposure, but interpretation still needs care. Programs often rely on defined cutoffs and treat ETG as a screening indicator that may require context and, in some cases, confirmation. Your policy should spell out how you handle incidental exposure claims and what documentation you expect.
“Does fentanyl on a cup catch all fentanyl-related risk?”
Not necessarily. Fentanyl screening depends on what the assay targets and the cutoff used. A cup can help you screen for fentanyl exposure, but it doesn’t replace clinical judgment, symptom assessment, or laboratory methods when you need definitive identification. If fentanyl risk is central to your program, pair screening with a clear confirmation pathway.
How to choose the right ETG/Fentanyl cup for a professional program
For professional use, choose based on your workflow and documentation needs—not just the number of panels. ETG and fentanyl often appear in programs focused on recency, relapse monitoring, safety-sensitive roles, or opioid risk. Those programs run better with consistent products and repeatable procedures.
Use these selection criteria to avoid mismatches
- Regulatory fit: verify FDA 510(k) clearance if your policy requires it.
- Workflow fit: pick a format your staff can run the same way every time.
- Result handling: decide what happens after a non-negative result before you run the first test.
- Documentation: keep package inserts, lot numbers, and written procedures organized and accessible.
- Program alignment: confirm the panel list matches what you actually need to monitor.
Optional FAQ
How do I ask a vendor the right question about FDA clearance?
Ask: “Do you have an FDA 510(k) clearance number for this exact cup and panel configuration, and can you share documentation that matches the product name?”
What should I do if a listing does not mention a 510(k) number?
Treat it as unverified. Ask for clearance documentation or choose a product with clear regulatory support that matches your requirements.
Should I treat a negative result as final?
Many programs accept a negative screening result for routine decisions. Your policy should still define when confirmation is required (for example, disputed results, unexpected findings, or high-risk clinical scenarios).
Conclusion
Multi-panel drug test cups that include ETG and fentanyl can work well in professional screening programs, but you can’t assume FDA clearance based on a category label or product title. Verify the regulatory status for the exact cup you’re buying, then support it with disciplined collection practices and a clear confirmation process for non-negative results. If you want one cup that covers alcohol exposure screening and fentanyl screening in a practical workflow, 12 Panel ETG & FEN Cup is the best choice from 12 Panel Now.