The Chemical and Biological Principles of Urine Drug Screening
When you are evaluating drug testing supplies for your clinic, workplace, or rehabilitation center, it is easy to view testing cups as simple plastic commodities. But behind every FDA-approved, CLIA-waived testing device is a highly sophisticated molecular engine. As a decision-maker in 2026, you aren’t just buying plastic—you are investing in chemical reliability.
You need to know that a presumptive positive is legally and scientifically defensible. Yet, when researching how these tests actually work, you are usually forced to choose between dense, impenetrable academic journals on PubMed or surface-level consumer blogs explaining “how to pass” a test.
Neither helps you make an informed purchasing decision.
This guide bridges that gap. We are going to decode the molecular “lock-and-key” mechanisms of modern immunoassays, explain the chemistry behind specimen validity, and provide the scientific bedrock you need to confidently evaluate rapid urine screening solutions.
The Molecular Engine: Why We Target Metabolites
To understand how a 99% reliable 12-panel drug test functions, we first have to look at what the test is actually searching for. Rapid urine screens rarely look for the parent drug itself. Instead, they hunt for metabolites—the chemical byproducts created after the human liver processes a substance.
Why target metabolites? Because parent drugs metabolize too quickly to offer a reliable detection window.
Consider marijuana. If a test only looked for active Delta-9 THC, the detection window would be incredibly narrow. Instead, our testing cups are engineered to detect THC-COOH (11-nor-9-carboxy-THC), a lipid-soluble metabolite that remains in the system for up to 30 days.
The mechanism driving this detection is the immunoassay. At the bottom of each testing strip are microscopic antibodies designed to act as biological “locks.” These antibodies are calibrated to bind perfectly with specific drug haptens (the “keys”). This binding isn’t magic; it is driven by precise intermolecular dynamics, primarily a combination of hydrogen bonds, electrostatic forces, and Van der Waals interactions. When the molecular shape and charge align perfectly, the antibody binds to the metabolite.
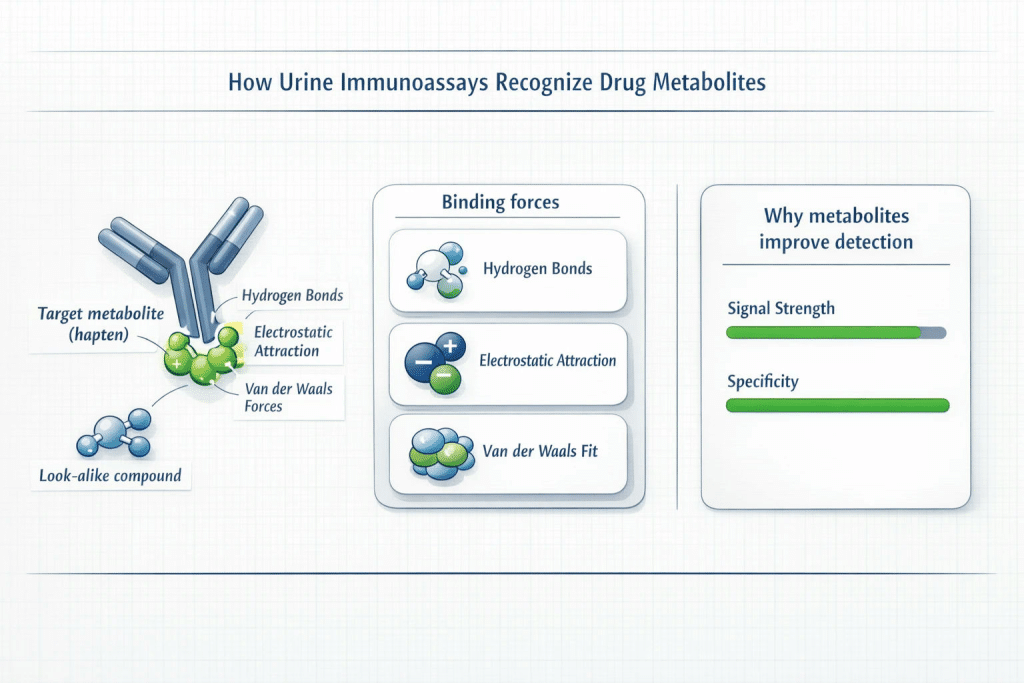

The Competitive Binding Paradox: Reading the Lines
One of the most frequent questions we hear from administrators onboarding new staff is: “Why does a missing line mean the test is positive?”
It seems counterintuitive. In almost every other rapid diagnostic test (like a COVID-19 or pregnancy test), the appearance of a line indicates a positive result. However, lateral flow drug testing cups rely on a principle called Competitive Binding.
Here is how the chemistry unfolds when a urine sample enters the test strip:
- The Dye Conjugate: As the urine travels up the absorbent strip, it picks up specialized, dye-labeled antibodies.
- The Race to the Test Line: Farther up the strip at the “Test” (T) region, synthetic drug proteins are permanently anchored to the membrane.
- The Negative Scenario: If the urine is clean, those traveling dye-labeled antibodies have nothing to bind to in the liquid. So, they continue up the strip, hit the anchored drug proteins, bind to them, and deposit their dye. A line appears, indicating a negative result.
- The Positive Scenario: If drug metabolites are present in the urine, the dye-labeled antibodies immediately bind to the metabolites while in transit. By the time they reach the Test region, their binding sites are full. They cannot bind to the anchored proteins, so they wash right past them. No line appears, indicating a presumptive positive result.
A missing line isn’t a failure of the test; it is the visual proof of a successful chemical block.
Chemical Interference and Cross-Reactivity
No discussion of molecular testing principles is complete without addressing false positives. While modern Enzyme Multiplied Immunoassay Techniques (EMIT) and similar screens boast specificity rates exceeding 99%, the universe of chemistry is vast, and structural “lookalikes” do exist.
When a false positive occurs, it is rarely due to a defective cup. It is usually the result of cross-reactivity—a perfectly normal chemical reaction where a legal medication’s molecular structure closely mimics a scheduled drug.
For example, the antidepressant Wellbutrin (bupropion) and the antihistamine Benadryl (diphenhydramine) possess chemical architectures that can occasionally trigger the amphetamine or methadone antibodies, respectively. Similarly, certain benzodiazepines (like Lorazepam) can sometimes exhibit sensitivity drops—acting stealthily and requiring enzymatic hydrolysis to be fully unmasked in a laboratory setting, leading to occasional false negatives on basic screens if concentration levels are exceptionally low.

The Screening vs. Confirmation Workflow
Because of these chemical lookalikes, high-performing facilities utilize a two-tiered evaluation workflow:
- Rapid Screening: Cost-effective, CLIA-waived 12-panel cups filter out the definitively negative samples instantly. At an entry point of just $1.69 per cup, this clears the vast majority of your testing volume economically.
- Lab Confirmation: Any presumptive positive is sent for GC-MS (Gas Chromatography-Mass Spectrometry) or LC-MS testing. Mass spectrometry doesn’t rely on antibodies; it shatters the molecule and weighs its individual atoms, providing an irrefutable chemical fingerprint.

Specimen Validity Chemistry: Catching Adulteration
As the stakes for passing drug screens remain high, the methods used to subvert them have grown more sophisticated. Evaluating a urine test today requires understanding Specimen Validity Testing (SVT)—the chemistry used to differentiate a genuine human sample from synthetic or adulterated urine.
Modern multi-panel cups are integrated with SVT strips that react to specific chemical thresholds:
- Creatinine: A natural byproduct of muscle breakdown. If creatinine levels fall below 20 mg/dL, it suggests severe dilution (flushing).
- Specific Gravity: Measures the density of urine compared to water. A low specific gravity confirms the dilution suggested by low creatinine.
- pH Levels: Normal human urine sits between 4.5 and 8.0. Household chemicals like bleach or ammonia, often used to destroy drug metabolites, will spike or crash the pH wildly outside this range.
- Nitrites/Oxidants: Commercial adulterants designed to oxidize THC metabolites leave behind a chemical footprint. High nitrite levels in a sample without a corresponding UTI are a massive red flag.
By 2026, clinical technology has advanced even further to counter synthetic urine (like U-Pass). Premium validity testing can now look for urinary tract glycoproteins (UTP-II & III) which are biologically impossible for synthetic manufacturers to replicate. Including SVT metrics directly on your testing cup secures the chain of custody and validates the molecular integrity of the result before you even look at the drug panels.
The Limits of Urine Toxicology
While understanding what urine toxicology does is vital, knowing what it cannot do is equally important for resource allocation.
Urine drug immunoassays are solely designed to detect exogenous chemical compounds and their metabolites. They cannot be used for Preimplantation Genetic Testing for Aneuploidies (PGT-A), nor are they Nucleic Acid Amplification Tests (NAAT) used for diagnosing sexually transmitted infections. A toxicology screen measures historical chemical intake—it does not map DNA, sequence viruses, or gauge current in-the-moment impairment like a blood test or breathalyzer would.
By utilizing rapid cups exclusively for their intended purpose—detecting historical substance use with 99% accuracy—you optimize both your budget and clinical workflow.
Frequently Asked Questions (FAQ)
Are rapid urine tests as accurate as laboratory tests?
They serve different functions in the same ecosystem. Our CLIA-waived, FDA-approved cups offer a 99% reliability rate for screening—meaning they are incredibly accurate at proving a negative result and identifying presumptive positives. However, laboratory mass spectrometry is required to legally confirm the exact molecular weight of the substance and rule out cross-reactivity.
Why do you test for THC-COOH instead of active THC?
Active THC metabolizes out of the human body in a matter of hours, making it nearly impossible to catch in a standard testing window. THC-COOH is a lipid-soluble metabolite that stores in fat cells, allowing our tests to reliably detect marijuana use up to 30 days after consumption.
Does a faint line mean a low level of drugs in the system?
No. Because these cups use competitive binding, the presence of any line—no matter how faint—indicates a negative result. The darkness of the line simply relates to the amount of dye conjugate that attached to the test line, which can fluctuate based on the specific gravity or acidity of the urine sample.
How do modern tests detect synthetic urine?
Quality multi-panel tests utilize built-in Specimen Validity Testing (SVT) to measure creatinine, specific gravity, and pH levels. Synthetic urines struggle to perfectly mimic these dynamic human biomarkers, and advanced lab confirmations can now identify the total absence of human glycoproteins (UTP-II and III) in fake samples.
What is the shelf life of a 12-panel testing cup, and does chemical degradation affect accuracy?
When stored properly in a climate-controlled environment, our individually sealed cups generally have an 18 to 24-month shelf life. Exposing the tests to extreme heat or freezing temperatures can denature the antibodies on the test strips, compromising the “lock-and-key” binding process and leading to invalid results.
Securing Your Testing Ecosystem
Understanding the complex chemical ballet of hydrogen bonds, competitive binding, and molecular haptens shouldn’t require a PhD in biochemistry—but it should give you absolute confidence in the tools you use.
At 12 Panel Now, we engineer our drug testing supplies to meet these rigorous scientific standards while maintaining the accessibility and affordability that clinical directors and HR managers demand. From comprehensive 12-panel cups with built-in SVT starting as low as $1.69, to specialized dip cards and oral swabs, we provide the reliable molecular engines your organization needs to maintain safety, compliance, and clinical integrity.