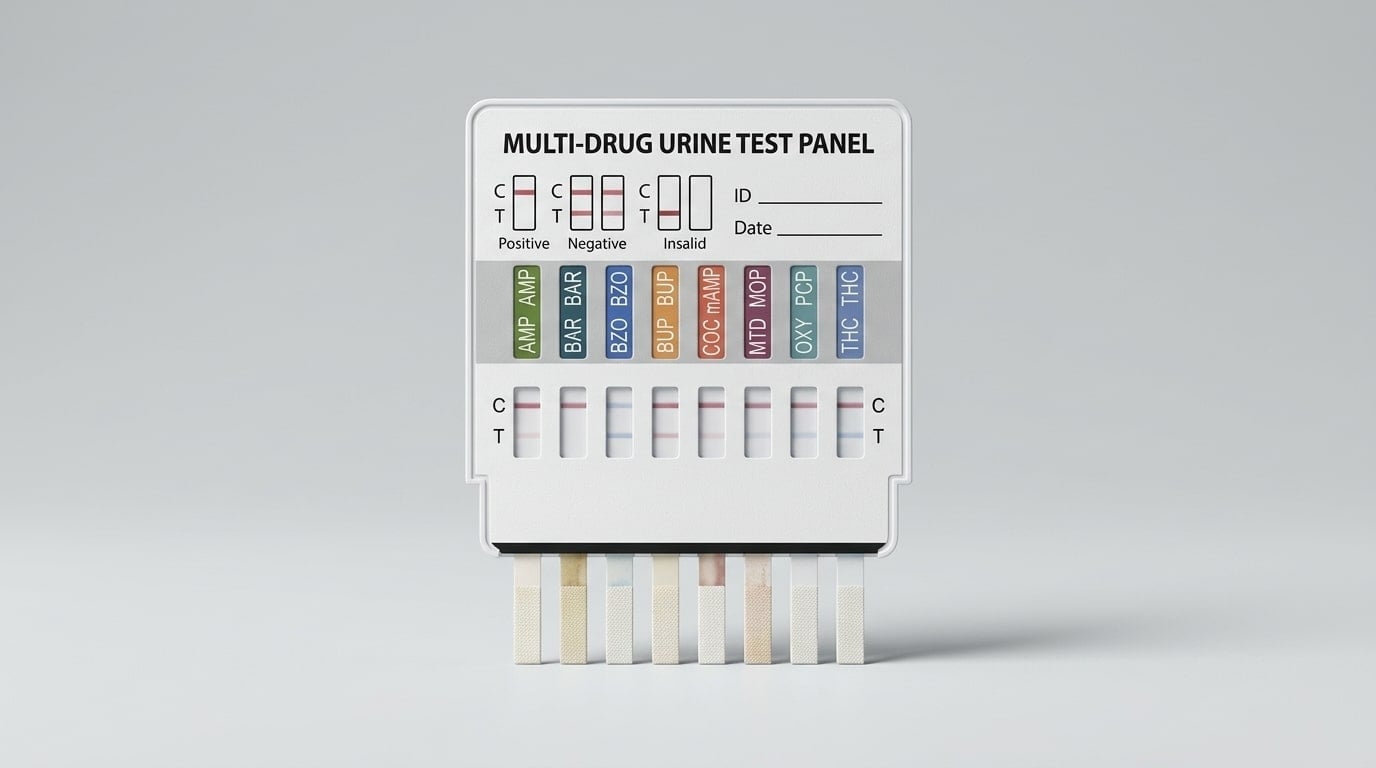
12 Panel vs 13 Panel vs 16 Panel Drug Test Cups Guide
Choosing a drug test cup gets confusing quickly. A 12 panel, 13 panel, and 16 panel cup each serve a different purpose, and the right choice depends on who you are testing, why you are testing, and which substances actually matter in your setting. This guide breaks down the differences so you can make a clear, confident decision.
What does the panel count really mean?
The panel count tells you how many drug categories the test screens for in a single sample. A higher number is not automatically better. It simply means the cup checks for more substances.
That distinction matters because every testing program has a different goal. An employer may want a broad workplace screen. A clinic may need to monitor both street drugs and prescription misuse. A parent may want a straightforward answer without paying for panels that add no real value.
When is a 12 panel drug test cup enough?
A 12 panel cup covers many common screening needs. It is a good fit when you want broad coverage of widely tested drug groups without paying for extras your program does not need.
Many buyers choose a 12 panel option for routine workplace screening, basic clinical use, school programs, or home monitoring. If your focus is on the substances most programs already watch for, a 12 panel cup is often the right middle ground.
Who should choose a 12 panel cup?
- Employers running pre-employment or random screening
- Clinics that need a fast first-line screen
- Probation or monitoring programs using standard drug lists
- Families who want clear at-home screening
A 12 panel cup makes sense when your testing policy is stable and your risk level is moderate. It gives you broad screening without adding panels your staff or records team then has to manage.
Why would someone move up to a 13 panel cup?
A 13 panel cup usually adds one extra substance that a 12 panel cup does not include. That may sound like a small difference, but it can matter if that added drug is showing up in your workplace, clinic population, or local community.
This is the key question buyers should ask: Which exact drug does the extra panel add? The answer depends on the product. One 13 panel cup may add a prescription drug category, while another adds a synthetic or region-specific concern. The value is in that specific addition, not in the number 13 itself.
When does a 13 panel cup make sense?
- Your policy requires one extra drug beyond a 12 panel screen
- Your area has a known issue with one added substance
- Your clinic monitors one additional misuse risk
- You want slightly broader coverage without moving to a much wider panel
If the extra drug is relevant, a 13 panel cup can be a smart upgrade. If it is not, you may just be adding cost and complexity.
What is the real value of a 16 panel drug test cup?
A 16 panel cup is designed for buyers who need wider screening. That usually means higher-risk settings, stricter monitoring, or programs that need to check for a broader mix of illicit and prescription substances.
A larger panel helps when standard screens leave important gaps. Treatment centers, pain management settings, probation programs, and some safety-sensitive employers may need that wider reach. A 16 panel cup lowers the chance that a substance goes unchecked simply because it was not included on a smaller test.
Who benefits most from a 16 panel cup?
- Rehab and recovery programs
- Pain management clinics
- Probation and court-ordered monitoring
- High-risk or safety-sensitive workplaces
- Programs addressing prescription drug misuse
The tradeoff is straightforward. A 16 panel cup gives you more information, but it also calls for tighter policy rules, clearer result interpretation, and a solid reason for screening a wider range of substances.
Is a higher panel count always better?
No. A higher panel count is only better when the extra panels match your actual testing goals. If you screen for drugs that do not matter in your setting, you add cost, paperwork, and the possibility of dealing with results your policy never planned for.
Some buyers assume more panels mean more protection. In practice, the better choice is the test that matches your real risk. The best test is the one that answers the right question, not the one with the biggest number on the box.

How should you choose between 12, 13, and 16 panels?
Start with the purpose of the test, not the panel count. Once you know why you are testing, the right range becomes much easier to identify.
Ask these questions first
- Why are you testing: hiring, compliance, treatment, monitoring, or home use?
- Which substances matter in your setting?
- Do you need a standard screen or a wider net?
- Will you need results fast on site?
- Do you have a plan for handling non-negative results?
A workplace with a standard policy may do very well with 12 panels. A program with one added concern may need 13. A clinical or court-driven setting may need 16. The right choice should reflect your risk level, policy, and budget.
What about accuracy and result speed?
Panel count is only one part of test quality. Buyers should also look at speed, ease of use, and whether the product meets recognized standards. In point-of-care testing, fast and clear screening matters because people often need answers at the time of collection.
That is why many professionals and individual buyers look for products that support on-site screening with simple handling and recognized clearance status. In many cases, a rapid urine dip card is a practical option when a full cup format is not necessary.
Should home users choose the same panel count as clinics or employers?
Usually not. Home users typically need a focused answer, not the broadest screen available. If you are testing a family member or checking for a specific concern, choose the panel range that matches that concern. At home, wider is not always more useful.
Clinics, employers, and probation programs operate under different pressures. They may need broader coverage for policy compliance, treatment oversight, or safety reasons. A home buyer is usually better served by relevance, simplicity, and easy interpretation.
What is the smartest choice for most buyers?
For many buyers, a 12 panel screen hits the sweet spot. It provides broad screening, supports quick decisions, and avoids the added reach that some programs simply do not need. That balance works well for many employers, clinics, and individual buyers who want practical coverage without making the process harder to manage.
If your program does not call for the added drug in a 13 panel cup or the wider spread of a 16 panel cup, a 12 panel option is usually the cleaner choice. It keeps your testing process focused and easier to run.
FAQ
Is a 13 panel test much better than a 12 panel test?
Not automatically. Its value depends on the one extra drug included. If that drug matters to your testing goal, then it may be worth it. If not, the extra panel may add very little.
Should I buy a 16 panel cup for workplace testing?
Only if your workplace policy or risk level calls for broader screening. Many employers do well with fewer panels. Safety-sensitive roles or higher-risk settings may justify more.
What is the best option for fast on-site screening?
A rapid point-of-care test with clear instructions and recognized compliance status is often a strong choice. Many buyers prefer a dip card or cup that delivers quick results without sending every sample to a lab first.
Conclusion
The right panel count depends on your reason for testing, the substances you need to screen for, and how simple you want the process to be. For many buyers, 12 panels offer the best balance of coverage and practicality. If you want a fast, reliable screening option, 12 Panel Dip Test – FDA approved and CLIA waived is a strong place to start from 12 Panel Now.