Instant Covid Test Boson, as low as $1.29 per test
$1.29 - $1.99
Authorized by FDA under an Emergency Use Authorization (EUA)
FREE SHIPPING 100 Covid test, same day shipping
| Covid test | |
| Nucleocapsid Protein Antigen | |
| Choose Quantity | Price per test |
For larger Orders call or text Alex 561-879-7737, John 561-879-7732 or Mona 561-879-7733
For all Shipping Inquiries call or text Adi 561-879-7735
Orders will only be shipped on business days before 3.00 pm EST. Friday overnight delivery will only be received on Monday.
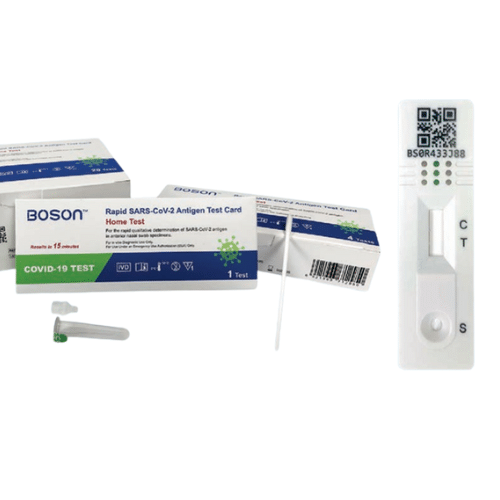
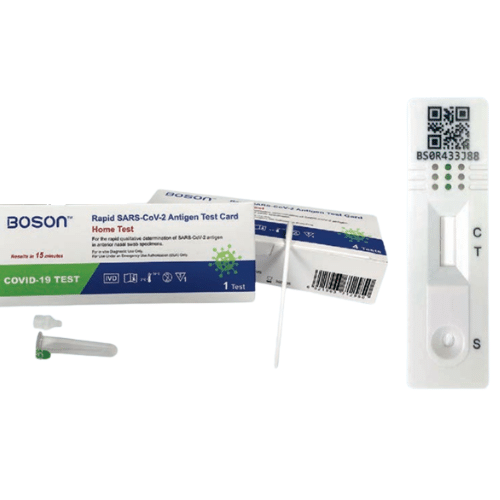
What is an Instant Covid Test Boson
The Instant Covid Test is a lateral flow test for detecting the nucleocapsid protein antigen from SARS-CoV-2 in a qualitative manner.
Self-collected anterior nasal swab samples from patients aged 15 years or older with symptoms of COVID-19 within the first 7 days of symptom onset are permitted for non-prescription home usage with this test.
Adult-collected anterior nasal (nares) swab samples from patients aged 2 years and older with COVID-19 symptoms within the first 7 days of symptom onset are also permitted for non-prescription home usage with this test.
SARS-CoV and SARS-CoV-2 are not distinguishable using the Instant Covid Test.
The Instant Covid Test Boson is authorized for non-prescription self-use and/or testing another individual aged 2 years or older by an adult lay user. The Instant Covid Test is only for use under the Food and Drug Administration’s Emergency Use Authorization.
How to Use an Instant Covid Test Boson – Procedures
The lateral flow immunoassay technology is used in the Instant Covid Test Boson.
This assay enables for the quick detection of SARS-CoV-2 nucleocapsid protein.
To begin the test, a self-collected anterior nares swab sample in adults aged 15 and older or a swab collected by a parent or guardian in individuals aged 2 to 14 is put into the Tube. The liquid in the tube interacts with the specimen, allowing the right viral antigens to be exposed to the test antibodies.
The liquid in the tube now containing the specimen is poured into the COVID-19 Test Card’s Sample Port.
Test Result Explanation

Swab
A non-invasive nasal swab should be brushed against the inner wall of both nostrils 5 times each in a circular motion.
Our nose swab is gentle, absorbent, and only requires ½ – ¾ inches of insertion to allow you to test yourself comfortably.

Dip
Insert the swab containing the sample into the tube’s bottom and agitate it 15 times in the fluid. Pull the swab out by squeezing the tube’s sides around it.

Drip
Put 3 drops of the mixed solution onto the sample port of the COVID-19 test card.
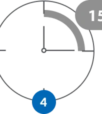
Wait 15 minutes
Start the timer. Your results
will be ready in 15 minutes.
Note: DO NOT interpret your test result until after your 15-min timer has completed, as the T line may take as long as 15 minutes to appear.
Reading the Results
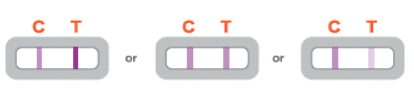
Positive Result
A POSITIVE result must show BOTH a C line and a T line. A positive result means that viral antigens from COVID-19 were detected and the individual is positive for COVID-19.

Negative Result
A NEGATIVE result will show ONLY a C line. A negative result means that viral antigens from COVID-19 were not detected and that the individual is presumed negative for COVID-19.

Invalid Result
The test is INVALID if there is no line or if there is only a T line. The test did not function properly, resulting in an invalid result. You’ll have to test again using a new test kit. If the test result is still invalid after retesting, contact your doctor or a local COVID-19 facility. An invalid result does not show whether the person had COVID-19 or not, and the test should be redone.
Conditions of Storage
Keep the Instant Covid Test Boson between 36 and 86 degrees Fahrenheit (2 and 30 degrees Celsius) in a dry place. Before using, make sure all test components are at room temperature, between 65 and 86 degrees Fahrenheit (18 and 30 degrees Celsius). The COVID-19 Test Card should be used within 1 hour of opening the foil pouch. Before the expiration date on the box, the Instant Covid Test Boson is stable.
Control of Quality
The device’s “control line (c)” contains a procedural internal control that is utilized to confirm that the applied specimen has moved well into the device.
It is coated with goat anti-rabbit IgG, and when the sample is introduced, a red colored line should appear.
What is the Instant Covid Test Boson and how does it work?
The Instant Covid Test Boson is an antigen test.
When COVID-19 is present in the body, the SARS-CoV-2 virus can be found in nasal secretions (the virus that causes COVID-19).
The Instant Covid Test Boson can detect microscopic components of the SARS-CoV-2 virus in your nasal secretions, known as N protein or antigens.
Do I have to wait 15 minutes to find out the outcome?
Yes. Do not validate your result until your 15-minute timer has expired after inserting the sample to the sample port. The positive indicating line could take up to 15 minutes to show, therefore if you don’t wait that long, you might get an inaccurate result (false negative).
Do I have to run two tests every time?
You can utilize one single test of the Instant Covid Test Boson if you have COVID-19 symptoms and are within the first 7 days of symptom onset. If you want, you can run two tests at the same time.
COVID-19 can be detected more immediately and the spread of infection can be reduced if you test more regularly.
If you don’t have COVID-19 symptoms or if you’ve had them for more than 7 days, you’ll need to do two tests spread out over two or three days, with at least 24 hours and no more than 48 hours between them.
Will the test hurt?
The nasal swab is not sharp, and it should not cause any discomfort. The swab can be a little uncomfortable or tickly at times.
If you experience any discomfort, please discontinue the test and seek medical help.
Is it possible to reuse the Instant Covid Test Boson?
No, the Instant Covid Test Boson’s components are not reusable and must be used just once.
How far should I insert the Nasal Swab?
Gently push the entire absorbent tip of the swab into your nostril (typically 1/2 to 3/4 of an inch).
When swabbing a child’s nostril, the greatest depth of insertion may be less than 34 of an inch, and you may require a second person to hold the child’s head while you swab.
How much liquid should I squeeze into the Test Card's sample port?
3 drops.
| Weight | N/A |
|---|---|
| Dimensions | N/A |
| As low As | As low As |
| Price | $1.29 |
You must be logged in to post a review.


All Orders Will Be Shipped the Same Day If Placed Before 3:00 PM EST.
Next-day Shipments Will Not Be Delivered on Weekends, and That the Time Taken on Air Shipments Should Be Counted as Business Days.
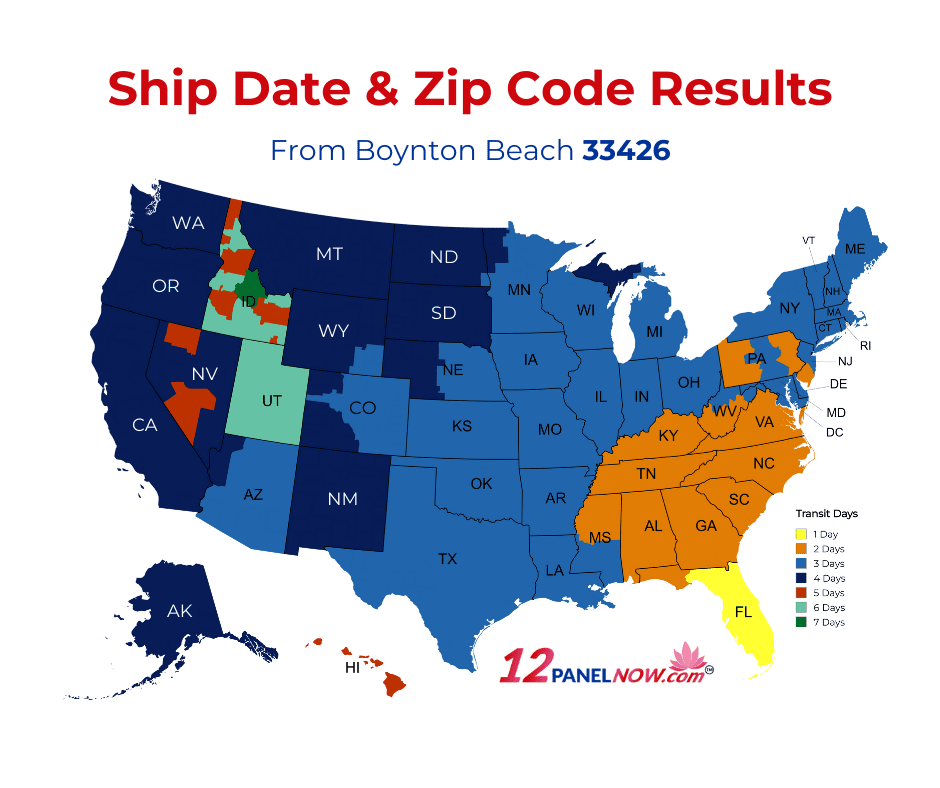
Why Does My Tracking Info Still Say "Label Created, Not yet in the System" When I Placed It X Days Ago?
If your order was placed via USPS, sometimes your label might not show up in their system even 1-3 days after they have received the package. The same can happen for UPS orders, although they almost always show up in their system the night which they are shipped. If you have any questions regarding the status of your order, please contact us and we will investigate the matter.
If I Pay for a next Day Air Delivery, Am I Guaranteed to Receive It the next Day?
Air deliveries performed by UPS are counted via business days, so you will receive your package the business day after it has been shipped. We also do not ship orders placed after 4 pm EST on the same day, so Next Day orders placed after 4 pm will be shipped the following business day.
Why Can't I Check the Tracking of My Package?
After you place your order, an email will be sent to you with the tracking number within a business day. If you did not receive this email, it's possible that the e-mail was entered incorrectly, sent to your spam folder, or blocked. If you did not receive this email, please call or email us and we'll be happy to give you the status of your shipment.
I Placed My Order but I No Longer Need It. Can I Return It and Get a Refund?
We accept returns for unused items as long as they are placed within 30 days after the day they are purchased. Although shipping is free upon order, we still incur a cost for every order that is shipped, and you will be refunded the amount of your order once we receive the returned goods, minus the cost of shipping.




































Reviews
There are no reviews yet.