Best Practices for Oral Fluid Collection & Sample Integrity
The shift toward oral fluid testing is one of the most defining trends in workplace safety and clinical screening today. If you are evaluating drug screening protocols in 2026, you already know the operational benefits. Oral fluid tests boast a refusal rate three times lower than traditional urine testing. They offer an unparalleled “recent impairment” window—detecting THC within a tight 4 to 24-hour timeframe, a precision metric urine testing simply cannot match.
But adopting oral testing and executing it flawlessly are two different challenges.
A screening program is only as strong as its sample integrity. Even the most highly rated, CLIA-waived testing devices rely entirely on the collector’s ability to secure a legally defensible, uncontaminated sample. We are going to bridge the gap between heavy federal regulatory guidelines and the practical, on-the-ground execution your team needs to run an airtight testing program.
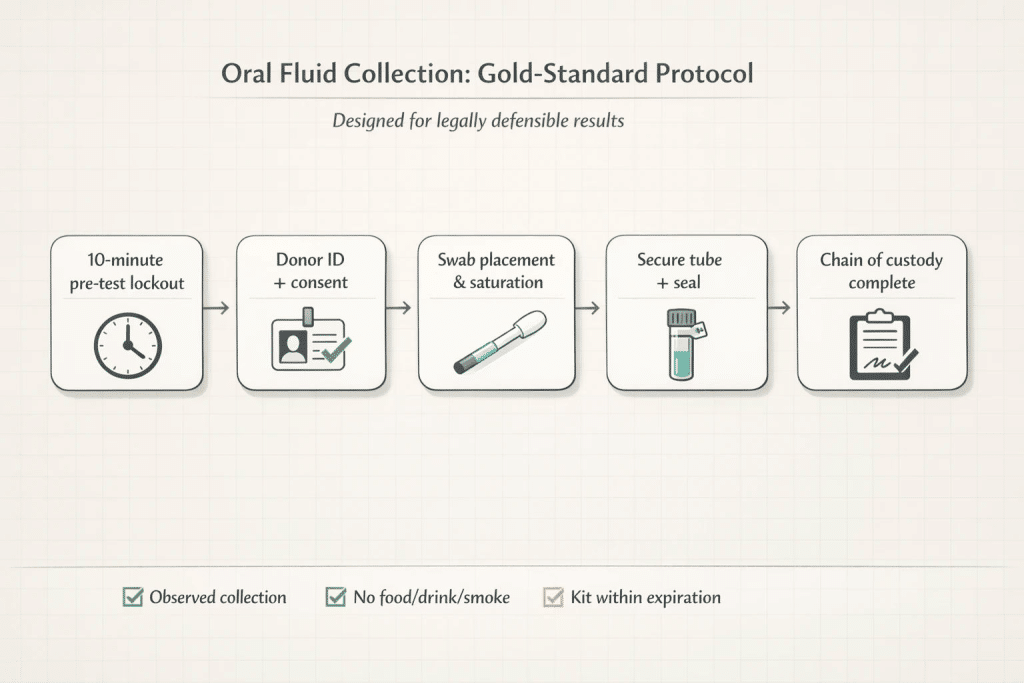
The Gold Standard Protocol: Step-by-Step Collection
Achieving compliance starts before the testing device even leaves its packaging. The standard operating procedure requires a clinical mindset, ensuring every step from observation to sealing builds a solid chain of custody.
1. The 10-Minute Pre-Test Lockout
This is the most critical and frequently overlooked step in oral screening. Before testing begins, the donor must undergo a strict 10-minute observation period. During this lockout window, the donor cannot eat, drink, chew gum, or use tobacco products. This ensures residual food particles or mouthwashes do not trigger false positives or invalidate the sample.
2. Device Administration and Saturation
Instruct the donor to pool saliva in their mouth before inserting the collection pad. Most premium testing supplies utilize a visual saturation indicator—a critical feature that removes the guesswork for your collectors. Whether your facility utilizes a standard multi-panel cup equivalent or a highly specific 1 black mouth swab test, the mechanism remains similar: the sponge must remain in the mouth until the indicator color changes, confirming adequate volume.
3. Securing the Split Specimen
For a test to be legally defensible, it must support split specimen testing. Once collected, the sample is often divided into Tube A (primary) and Tube B (secondary). Securing these with tamper-evident seals in front of the donor is non-negotiable. This guarantees that if a donor disputes a non-negative result, Tube B remains perfectly preserved for GC/MS or LC/MS confirmation testing.
Why Oral Swabs Fail (And How to Prevent It)
Even with premium supplies, user error happens. Understanding where collections break down allows you to train your staff proactively.

According to recent Q4 2024 industry data, roughly 2% to 5% of all oral fluid tests face rejection at the laboratory level. Drilling down into those rejections reveals a clear target for process improvement: 60% to 70% of failed tests are rejected due to “Insufficient Volume.”
When collectors rush the saturation phase or misinterpret the color indicator, the lab lacks the physical material required to run their assays. Investing in FDA-approved, high-reliability swabs with vivid saturation indicators directly mitigates this costly administrative failure.
The New Era of Validity: Handling Evolving Standards
Now that the workplace screening industry is fully operating under the stringent 2023–2025 SAMHSA updates, sample validity checks are more rigorous than ever. You can no longer just look at the saliva and assume it is testable.

Current validity standards require specific gravity, pH, and oxidant benchmarks to be strictly met. A valid oral fluid sample must maintain a pH balance between 5.0 and 9.0. Anything falling outside this window strongly indicates sample tampering. Modern screening tools are designed with these specific guardrails in mind, allowing collectors to flag adulteration instantly.
The Troubleshooting Vault: Dry Mouth and Adulteration
Real-world collections rarely happen under perfect conditions. The mark of a reliable screening program is how it handles edge cases without sacrificing chain of custody.

Solving Xerostomia (Dry Mouth)
Dry mouth is the primary cause of insufficient volume. While older protocols simply suggested “waiting 10 minutes,” clinical best practices now dictate a controlled hydration schedule. If a donor cannot produce adequate saliva:
- Provide a maximum of 8 ounces of water.
- Allow a brief rinse, but ensure the water is swallowed.
- Wait exactly 10 minutes to allow natural saliva regeneration before reintroducing the swab.
This prevents the sample from becoming diluted while physically enabling the donor to comply.
Identifying Adulteration
Because oral fluid testing is highly observable, adulteration is difficult but not impossible. Common attempts involve localized oxidants like hydrogen peroxide or swishing vinegar right before walking into a facility.
Your defense here relies entirely on the 10-minute lockout. Because saliva regenerates continuously, any external chemical introduced to the mouth washes away naturally within minutes. By enforcing the observation window, you effectively neutralize chemical adulteration before the swab enters the donor’s mouth.
Elevating Your Chain of Custody
As corporate screening policies mature, alignment with high-level protocols like a Securitas mouth swab program or OralTox-style indicators becomes a baseline expectation. These systems rely on rapid, visible results combined with a sealed laboratory-ready design.
For facilities processing high volumes of tests, the physical device must do half the work for you. Choosing solutions that lock tightly, clearly display negative and non-negative panels, and seamlessly fit into your shipping boxes prevents clerical errors from invalidating a perfectly collected sample.
Confident Decisions Drive Reliable Results
Building an airtight screening program means evaluating not just the protocol, but the tools your team relies on every day. By understanding the common pitfalls of oral fluid collection—from insufficient volume to complex validity checks—you position your organization to run highly efficient, deeply reliable tests.
Empower your collectors with straightforward procedures and equip them with testing supplies that meet the highest standards of reliability. When quality and affordability align, maintaining a safe, compliant environment becomes second nature.
Frequently Asked Questions
Are oral fluid tests legally defensible in 2026?
Yes. When collected following SAMHSA and DOT guidelines—specifically enforcing the 10-minute lockout and securing split specimens—oral fluid tests are highly defensible. Utilizing CLIA-waived and FDA-approved testing supplies further cements your program’s compliance.
Why choose oral fluid over traditional urine panels?
The primary advantage is candidate experience and risk mitigation. Oral testing removes the privacy concerns of bathroom collections, eliminating “shy bladder” scenarios. Furthermore, its narrow detection window (often up to 24 hours for substances like THC) is ideal for identifying recent use rather than historical, off-the-clock habits.
What should a collector do if the saturation indicator never changes?
If 15 minutes pass without the indicator activating, the collector should halt the test. Initiate the clinical dry mouth protocol—provide a small amount of water, wait an additional 10 minutes, and restart the test with a completely new, sterile swab.
How do we keep costs down while maintaining these high standards?
Quality does not have to drain your operational budget. By partnering with direct-to-customer suppliers who specialize strictly in testing equipment, organizations can secure high-reliability, multi-panel screening supplies at fractions of a dollar per test, maintaining both clinical accuracy and budget compliance.

