Configuring Multi-Panel & Single-Panel Urinalysis for Specific Detection Needs

If you are evaluating drug testing supplies for your organization right now, you already know the landscape has fundamentally shifted. Gone are the days when a basic 5-panel commodity cup was enough to ensure a safe, compliant workplace. Today, you aren’t just buying tests—you are engineering a strategic defense against an evolving array of synthetic threats, complex prescription abuses, and sophisticated evasion tactics.
For many decision-makers, the jump from standard panels to 12, 14, or 16-panel configurations creates decision paralysis. Does the incremental cost justify the coverage? How do you map specific panel configurations to your industry’s unique risk profile?
As a trusted partner to organizations nationwide, 12 Panel Now understands that you need more than just a list of products. You need a clear, authoritative framework to evaluate the exact configurations that will protect your operations while optimizing your testing budget.
The Evolution of Drug Screening: Moving Beyond the Basic 5-Panel
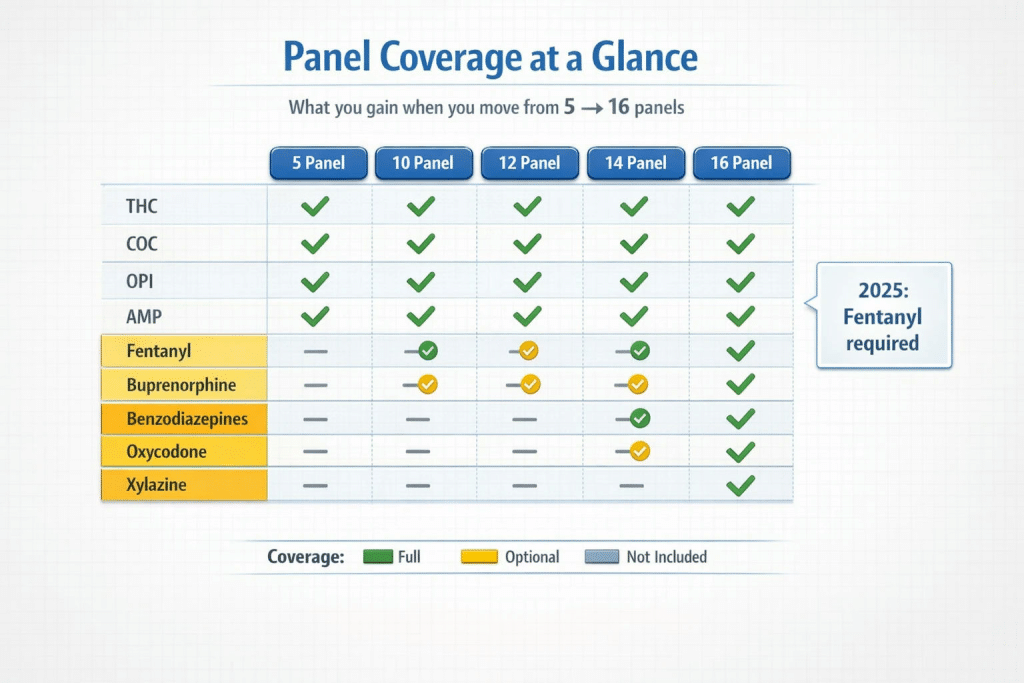
For decades, the standard 5-panel test was the industry baseline. However, recent data shows that 5-panel configurations miss up to 80% of modern synthetic threats. While they cover the basic “DOT 5,” they leave dangerous blind spots in today’s environment.
The turning point came with the HHS and Federal Register mandate requiring Fentanyl in federal workplace panels by July 2025. Now that we are well into 2026, Fentanyl screening is no longer an optional add-on—it is the baseline expectation for liability reduction. Evaluating which multi-panel configuration you need means looking at what substances are included at each tier, from 10 to 16 panels, and ensuring your choice aligns with current compliance standards.
Configuring by Use Case and Industry: A Strategic Blueprint
The most effective testing programs do not treat every employee and situation identically. Instead, they tailor the panel configuration to the specific intent of the test and the inherent risks of the industry.
Pre-Employment vs. Random Testing Dynamics
When screening new hires, cost-efficiency at volume is usually the primary driver. A standard 10-panel or 12-panel cup offers a robust safety net without breaking the budget. However, random testing requires a different approach. According to recent Quest Diagnostics Drug Testing Index data, Fentanyl positivity is seven times higher in random tests than in pre-employment screens.
This dramatic variance dictates a clear strategy: use affordable 10 or 12-panel cups for your high-volume pre-employment funnel, but deploy comprehensive 14+ panel tests for random screenings, post-accident investigations, and reasonable suspicion cases where the likelihood of detecting high-risk synthetics spikes.
The Adderall Nuance in Healthcare and Corporate
In healthcare, pharmacy, and high-stress corporate environments, the abuse of prescription medications like Adderall or Buprenorphine is a growing concern. Evaluating test results in these sectors requires careful nuance. A comprehensive multi-panel test will flag amphetamines, prompting a crucial dialogue between HR and the employee regarding legitimate prescription disclosure versus illicit abuse. Choosing a panel that accurately isolates these compounds prevents false accusations while maintaining rigorous safety standards.

The “Cheater-Proof” Configuration: Defeating Powdered Urine
Perhaps the biggest unstated fear during the evaluation process is test evasion. Search volume for “powdered urine” and synthetic masking agents has surged, meaning your testing program is only as good as its ability to detect adulteration.
A high-quality multi-panel cup isn’t just looking for drugs; it is verifying the integrity of the sample itself. This is where Validity Tests—which measure pH, Specific Gravity, Creatinine, and Nitrites—become your primary shield.
If a donor attempts to dilute their sample with water, the Specific Gravity and Creatinine levels will plummet, instantly flagging the cup. If they introduce chemical oxidants to mask THC or other substances, the pH and Nitrite indicators will expose the tampering. When evaluating your options, integrated adulterant testing is the critical trust signal that elevates a simple cup into a foolproof diagnostic tool.

Single-Panel vs. Multi-Panel Logic: Optimizing Your Cost-to-Benefit Ratio
One of the most common inefficiencies in corporate testing programs is using the wrong format for the wrong scenario. While competitors often treat single-panel strips as an afterthought, sophisticated administrators use them as highly targeted verification tools.
When to use Multi-Panel Cups:
Multi-panel cups (ranging from 10 to 16 panels) are your broad-spectrum screening tools. They are designed for initial pre-employment intakes, widespread random testing programs, and baseline post-accident reports. At pricing as low as $1.69 per cup, 12 Panel Now makes it financially viable to use comprehensive multi-panels as your organizational standard.
When to use Single-Panel Strips or Dip Cards:
Single-panel strips shine in monitoring, follow-up, and highly specific suspicion scenarios. For instance, if an employee is on a return-to-duty program following a previous positive THC result, you do not need to waste resources on a 14-panel cup every week. A highly accurate single THC strip acts as a cost-effective detox monitor. Similarly, single Fentanyl strips can be deployed rapidly if a specific hazard is suspected on a job site.

The Non-Negotiable: Why CLIA-Waived Matters
As you finalize your evaluation criteria, one technical specification stands above the rest: CLIA-waived status. Clinical Laboratory Improvement Amendments (CLIA) regulations ensure that point-of-care tests are simple to use and carry an insignificantly low risk of yielding incorrect results.
Using CLIA-waived, FDA-approved products is what bridges the gap between “at-home” testing and lab-grade accuracy. It guarantees a 99% reliability rate, ensuring that your HR and safety teams can make legally defensible employment decisions with absolute confidence.
Frequently Asked Questions (FAQ)
Is the incremental cost of moving from a 10-panel to a 14-panel cup worth it?
Yes, particularly for random screening and post-accident testing. The slight increase in unit cost buys you coverage for high-risk substances like Fentanyl, Buprenorphine, and Xylazine—drugs that are driving modern workplace safety incidents but are entirely missed by 10-panel configurations.
How do I know if an employee is using synthetic or powdered urine?
You must select a cup equipped with built-in validity testing. These panels test the urine’s Specific Gravity, Creatinine, and pH levels. Synthetic urine or diluted samples will fall outside the normal human physiological ranges for these markers, immediately flagging the test as invalid or adulterated.
Why shouldn’t I just use single-panel tests to save money?
Single-panel tests are highly cost-effective for targeted monitoring (like return-to-work programs), but they leave massive blind spots if used for general screening. Multi-panel cups consolidate 10 to 16 tests into one rapid process, providing comprehensive risk mitigation for just a few dollars per cup.
Are rapid tests legally defensible in 2026?
Yes, provided they are CLIA-waived and FDA-approved. These certifications ensure a 99% accuracy rate. However, standard protocol dictates that any non-negative rapid result should be sent to a laboratory for GC/MS (Gas Chromatography/Mass Spectrometry) confirmation before taking adverse employment action.
Moving Forward with Confidence
Selecting the right urinalysis configuration isn’t about buying the most expensive cup—it is about matching the exact technical specifications of a test to your organization’s compliance requirements, budget, and risk profile. By combining broad multi-panel screens for general safety with targeted single-panels for verification, and insisting on built-in adulterant defense, you future-proof your program against an evolving landscape.
At 12 Panel Now, we blend family-owned service with clinical-grade reliability to deliver CLIA-waived solutions that empower your decision-making. Ready to configure the perfect testing protocol for your team? Explore our comprehensive ecosystem of multi-panel cups and single strips today, and secure the clarity your organization deserves.

